Lower limb support AM-OSK-Z/S-A


 Knee brace
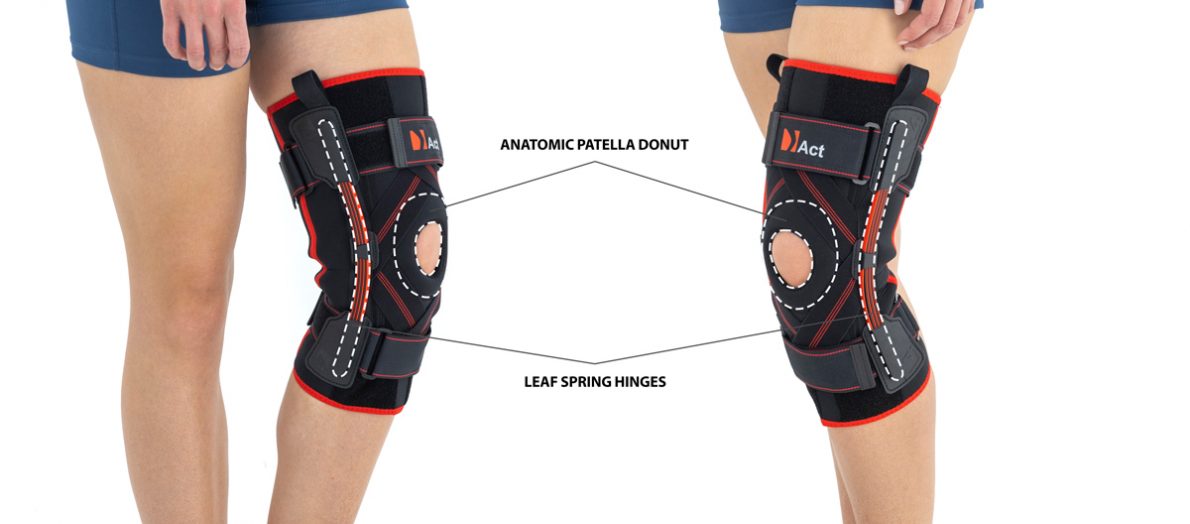
Knee brace Anatomic patella donut
Anatomic patella donut Class I medical device
Class I medical device Compression
Compression Leaf springs
Leaf springs Ligament protection
Ligament protectionAct
ACL KNEE BRACE WITH LEAF SPRING HINGES
Description
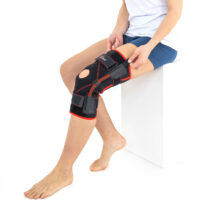
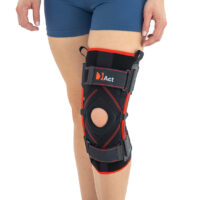
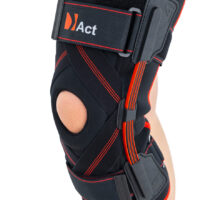
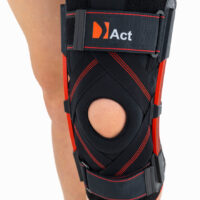
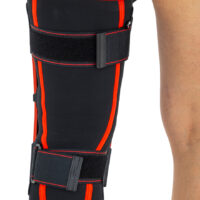
Closed knee brace AM-OSK-Z/S-A is a professional first class, medical product improving efficiency of walk, used in rehabilitation after damage of the knee joint. It is made of ActivePren™ and AirRubber III™.
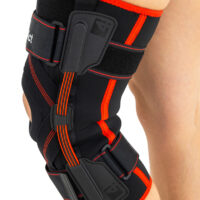
The brace is equipped with elastic splints, which stabilize knee in frontal plane. This splints are sides leaf spring hinges, which reflects physiological knee motion precisely. Additionally, the brace has independent sets of VELCRO tapes, frontal ACL support and patella support with antimycotic properties. The Impact brace is the best solution for active people (included athletes), who need frontal knee stabilization, maintaining the anatomic range of motion.
The brace is equipped with leaf spring hinges. The splints are made of high quality alloy of metal and plastic. This construction provides anatomic knee motion with excellent lateral knee stabilization. The AM-OSK-Z/S-A brace helps in knee extension, because of used high quality spring steel.
Purpose of use
AM-OSK-Z/S-A brace should be applied in cases of:
- knee dislocation,
- knee twist and sprain,
- side instability of the knee joint,
- knee ligaments LCL, MCL and ACL injuries,
- knee ligaments reconstruction,
- other surgeries (orthopedic),
- kneecap instability – patellar subluxation.
Sizes
| Size | Knee circumference | How to measure |
| M | 34-38 cm (13,4″-15″) |
 |
| L | 38-42 cm (15″-16,5″) |
|
| XL | 42-46 cm (16,5″-18,1″) |
Total length of the product: 35 cm (13,8″)
Gallery
Technology
MATERIALS
ActivePren™
ActivePren™ is an active three-layer material consisting of two elastic jersey cover fabrics and a core made of neoprene foam. This material is characterized by softness and high flexibility. A very important advantage of this material is the fact that it is not a knitted product, it does not have thick fibers, so that the weaves of the material do not imprint on the patient's skin and do not cause abrasionsin places of high compression. Products made of ActivePren are the strongest and most effective stabilizing orthoses available on the market.
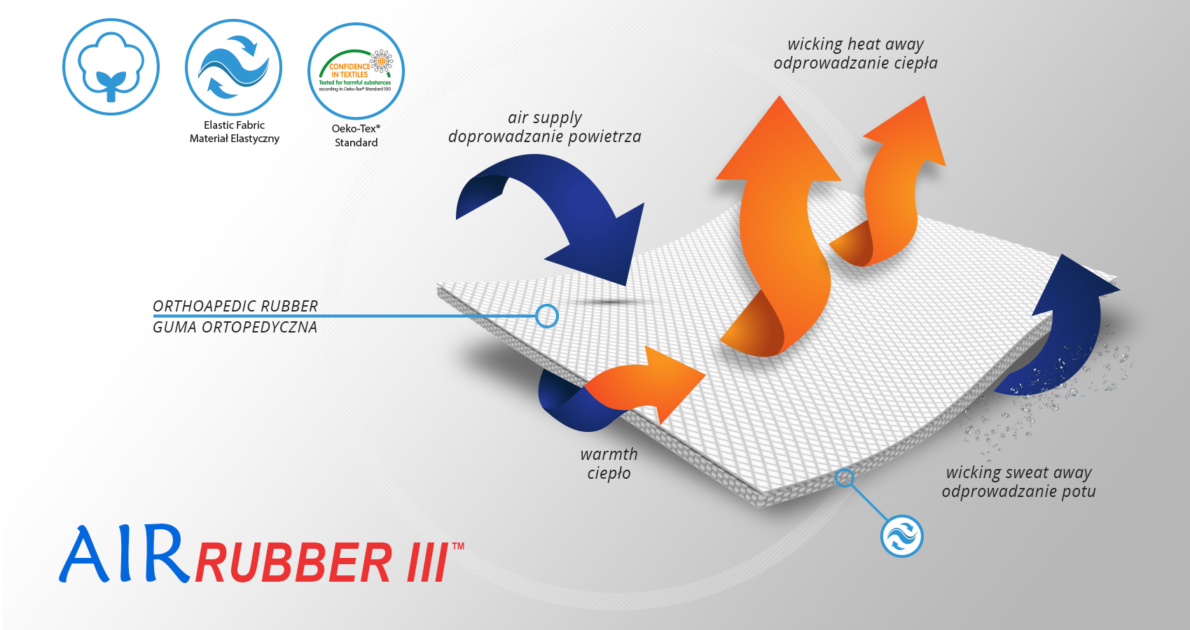
AirRubber III™
AirRubber III™ has unidirectional elasticity. It can be stretched, increasing the length, not width, what improves compression. Between the braids air flows freely and skin can breathe. In addition in high compression, these rubber braids provide a gentle massage for skin. Our orthopedic rubber is very friendly for skin.
STIFFENINGS
Leaf spring hinges
Leaf spring hinges are made of high quality alloy of metal and plastic. This construction provides anatomic knee motion with excellent lateral knee stabilization.

PADDINGS
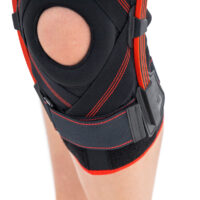
Patella stabilizers
Relief stabilizers of various shapes made in 2D technology. They are made of relieving foams connected on one side with a fabric friendly to the patient's skin, and on the other with a gripper, thanks to which the stabilizer can be attached to the adhesive element of the orthosis. Thanks to such a structure, these pads do not have to be sewn into the orthosis, and they can simply be fastened to it from the inside. These elements have an anatomical shape and are made of comfortable foam with proper hardness and elasticity, which guarantees the proper therapeutic effect.
Setting up
Downloads
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.