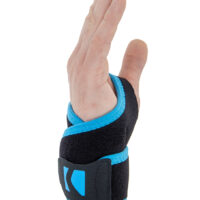
Wrist support U-SN


 Wrist brace
Wrist brace Class I medical device
Class I medical device Double-sided
Double-sided Latex-free
Latex-free Skin-friendly
Skin-friendly Universal size
Universal size Waterproof
WaterproofUNIVERSAL WRIST BRACE
Description
Wrist support made of UniPren™.
Applying
The support presses the muscles improving blood circulation and positively influencing the oxygen distribution through the whole body. This support provides perfect stabilization of the wrist, reduction of pain caused by overstress, old injuries and degenerative changes, increases the strength of the grip (e.g. tennis, weights lifting). Maintenance of stable temperature and elastic pressure results in reduction of swelling, helps in healing of post-injury effusions and hematoma. Enables faster come back to wok and training.
Purpose of use
- after wrist injuries
- bursitis
- joint degeneration or inflammation
Sizes
| Size | Wrist circumference | How to measure |
| Single size | min 13 cm – max 20 cm (min 5,1″ – max 7,9″) |
 |
Total height of the product: 10 cm (3,9″)
Gallery
Technology
MATERIALS
UniPren™
UniPren™ is a universal 3-layer material consisting of an external elastic polyamide cover knit with a self-adhesive function, an internal neoprene foam core and an elastic jersey cover knit. This material is characterized by softness and very high flexibility. A very important advantage of this material is the fact that it is not a knitted product, it does not have thick fibers, so that the weaves of the material do not imprint on the patient's skin and do not cause abrasionsin places of high compression. Products made of UniPren™ are the strongest and most effective stabilizing orthoses available on the market. Self-adhesive function, the raw material makes it much easier to use.
Setting up
Downloads
Accessories
ACCESSORIES / PRODUCTS TO BE USED WITH
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.