Finger splint AM-D-03


 Finger brace
Finger brace 4 in 1
4 in 1 Breathable
Breathable Class I medical device
Class I medical device Latex-free
Latex-free Recommended by specialists
Recommended by specialists Skin-friendly
Skin-friendlyFINGERS SPLINT
- Description
- Sizes
- Gallery
- Technology
- Setting up
- Downloads
- Accessories
Description
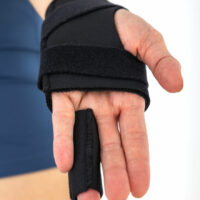
Hand brace to stiffen four fingers excluding the thumb.
The design of the product allows the patient or physician to choose which finger is eligible for immobilization. Each time, the brace can lock from one to four fingers.
The brace has the shape of a short glove with finger channels for finger-splints.
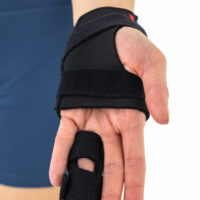
The rails are made of aluminum and are protected against the harmful effects of sweat by powder paint. The material for the liners has been chosen so that it was easy to make a desired shape by the physician.
The equipment of the brace are finger splints in open or closed versions. Each of the splints has a locking band. As standard the brace is equipped with one finger’s splint in the closed version.
In the product there is a thumb notch and installed peripheral safety belt.
The brace is made of stiffened velours knitwear and elastic Lycra.
Additionally, it is equipped with a lateral fastening strap increasing fitting. Moreover, short design of the brace does not limit the movement of the wrist.
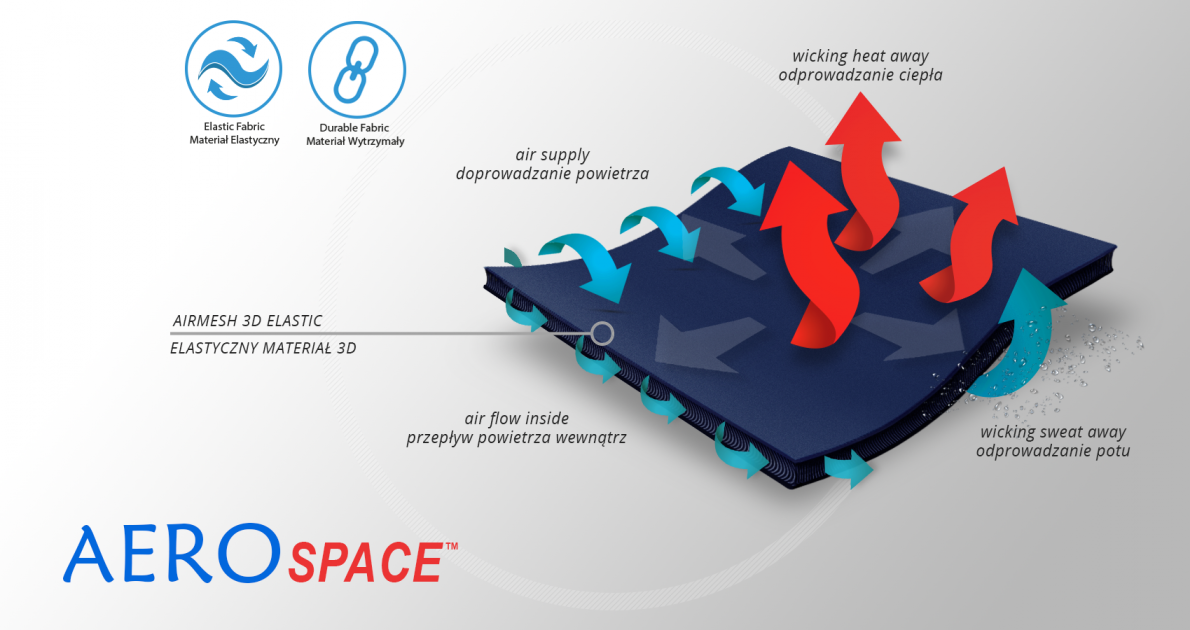
The brace is made of AeroSpace™.
Purpose of use
– II-V fingers fractures
– Injuries of collateral ligaments of MCP joints
– II-V fingers dislocations
– Rheumatic diseases of II-V fingers
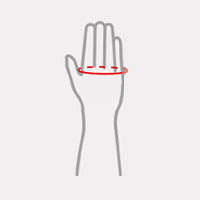
Sizes
Gallery
Technology
Setting up
Downloads
Accessories
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.