Forearm support AM-OSN-L-01


 Wrist brace
Wrist brace Anatomic wrist splint
Anatomic wrist splint Cast replacement
Cast replacement Class I medical device
Class I medical device ER
ER Finger separator
Finger separator Innovative
Innovative Recommended by specialists
Recommended by specialists Waterproof
WaterproofLONG OPEN FOREARM AND WRIST BRACE WITH FINGERS SEPERATION
Description
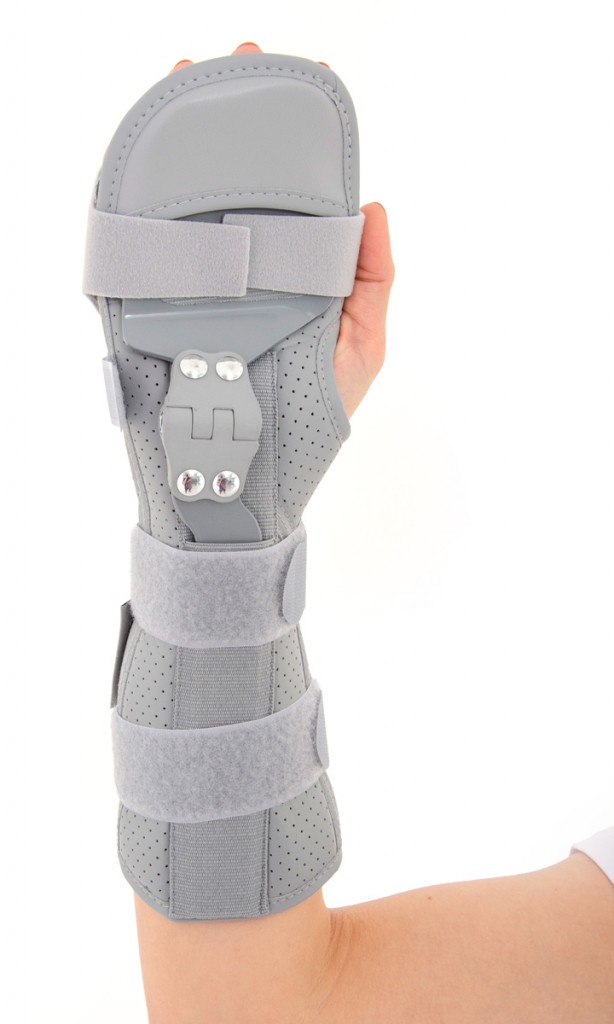
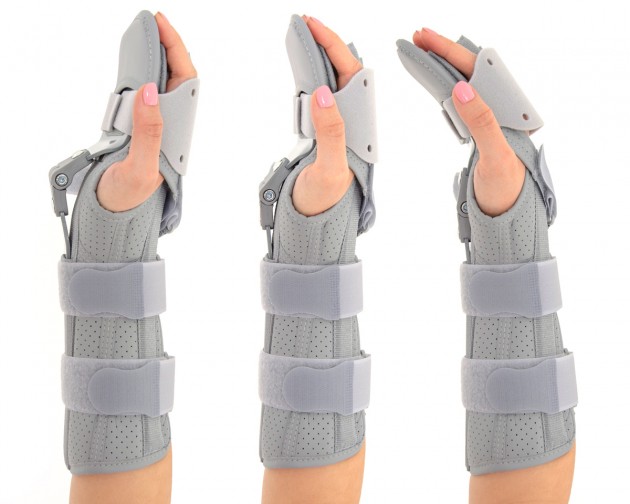
Long open forearm and wrist support was developed according to anatomical shape providing stabilization on the wrist and radiocarpal joint.
Wrap around design of brace allows easy-fitting and offers whole range of adjustment.
Dorsal Band on Fingers provides support on phalangeal area. The product is supplied with soft fingers separator, which prevents to fingers overlapping, anti-spasticity, relieves pain fingers and keeps them in correct position.
The orthosis is made of innovative fabric AirSanmed II™.
The palmar part of orthosis and forearm is equipped with anatomical shaped, malleable and removable strip with sweat resistant powder coating applied.
The fitting on the wrist is obtained by three sets of velcro straps, which ends of closure are provided with Plastic Covers, specially developed for patients with limited dexterity, such as osteoarthritis and wrist instability.
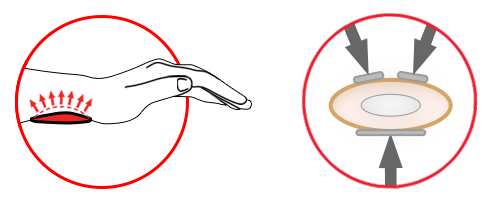
3 points stabilization
L3P (LOCK 3 POINTS)
LOCK 3-POINTS Circumferential Compression allowing adequate support and unloading of radiocarpal joints. The orthosis is provided with anatomically shaped, malleable and removable metal Strip for Palm and Forearm (sweat resistant coating) and 2 Dorsal plastic stays.
Indications
– Radiocarpal joint dislocation
– Tendonitis and Tendon strains, Tenosynovitis
– Carpal Tunnel Syndrome
– Rheumatoid Arthritis
– Overuse injuries
– Post-traumatic and postoperative (orthopaedic or rheumatoid)
– Central or Peripheral neurological injuries
– Pathologies requiring immobilization of wrist
Sizes
| Size | Wrist circumference | How to measure |
| S | 13-15 cm (5,1″-5,9″) |
 |
| M | 15-17 cm (6,1″-6,7″) |
|
| L | 17-19 cm (6,9″-7,5″) |
|
| XL | 19-21 cm (7,7″-8,3″) |
| Size | The length of the orthosis |
| S | 30 cm (11,8″) |
| M | 35 cm (13,8″) |
| L | 38 cm (15″) |
| XL |
Left/right side available.
Gallery
Technology
MATERIALS
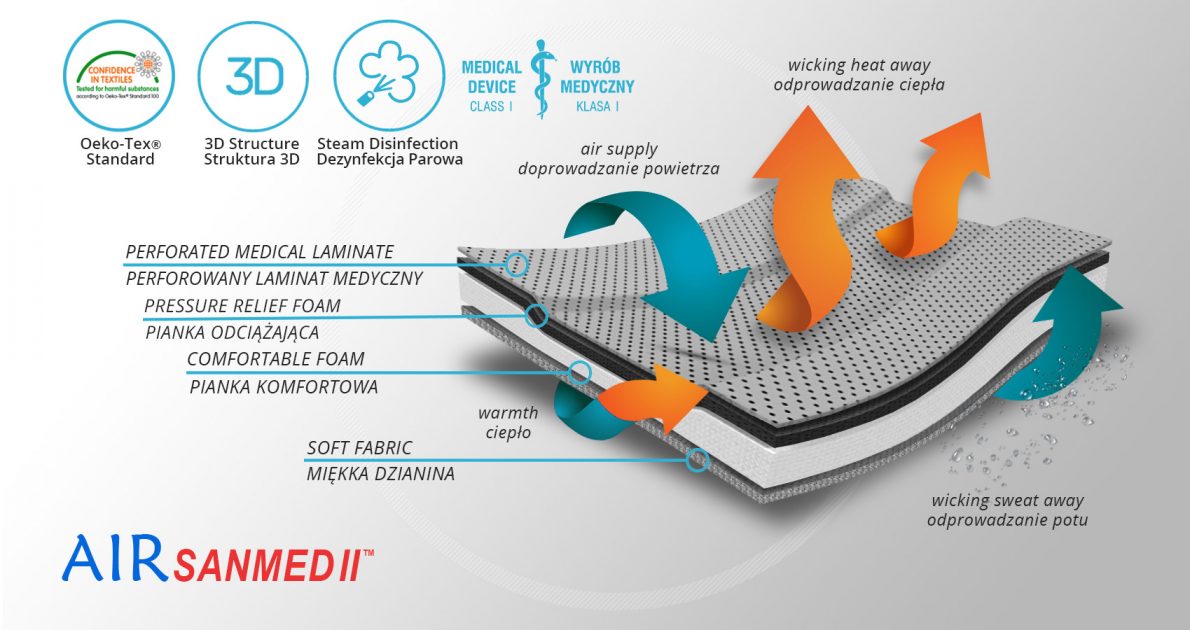
AirSanmed II™
AirSanmed II™ is a multi layered intelligent orthopedic textile which is perfect for orthopedic braces production. It helps to maintain its functionality and high comfort of use. External vapour barrier is washable and possible to chemical or vapour disinfection. It is perforated and laminated with off loading foam. Due to this fact, the fabric is breathable and reduces the pressure of aluminum stays and splints. Off-loading foam is connected with soft, comfort foam improving the comfort of use. It’s laminated with soft, breathable cotton with pH neutral for human’s skin. This knitted cotton is certified with OekoTex Standard100 and fully skin-friendly.
| ALLOWABLE DISINFECTANTS !!! | ||
| Type of active agent or surface-active agent | Maximum content in disinfectant | Spray the surface of the device with disinfectant, leave it for disinfectant’s activation, then wash the device and dry it. |
| 2-propane | 35% | |
| 1-propane | 25% | |
| Ethanol | 10% | |
| Alkyl Dimethyl Benzyl Ammonium Chloride | 0,2% | |
| Glucoprotamine | 25% | |
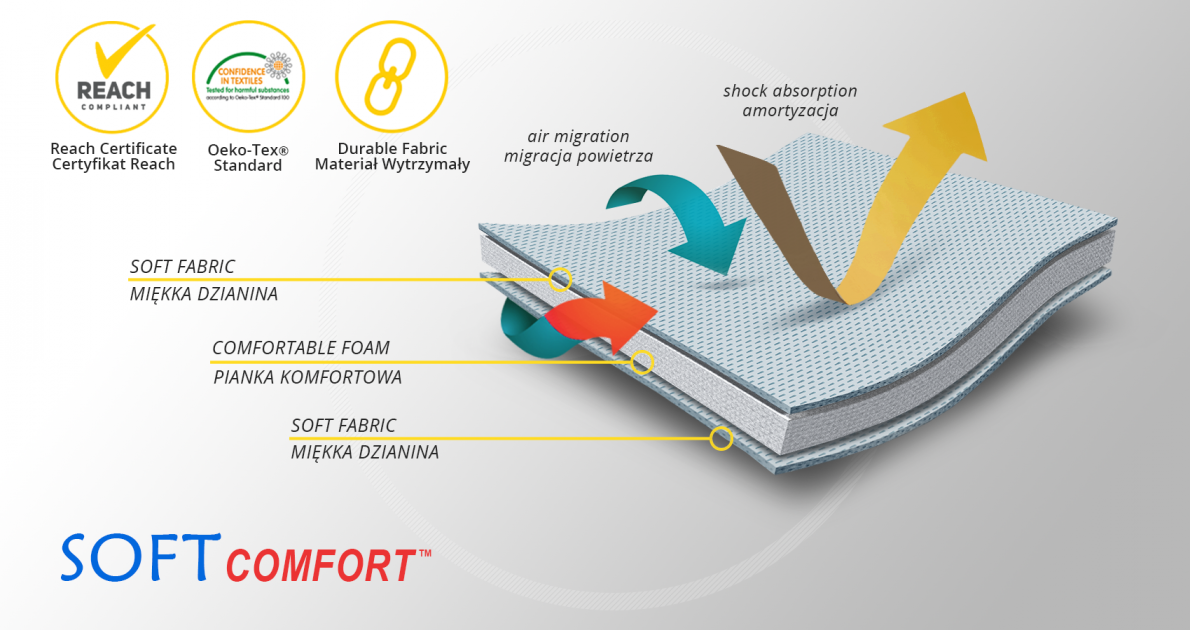
SoftComfort™
SoftComfort™ - a material specially designed to relieve very delicate places on the patient's body. The cover knit used in the production is made in such a way that it can come into contact with very sensitive skin. This raw material has the Oeko-Tex Standard 100 certificate and complies with the European REACH standard. The structure of the material consists of a very soft comfort foam laminated with a delicate cover knit. The fabric has a perforation that facilitates the flow of air through the material and allows the skin to breathe. The SoftComfort™ material is perfect for the production of linings relieving the rigid and inflexible elements of orthopedic orthoses.
STIFFENINGS
Anatomically shaped hand splint
Plastic stays
They come in various widths and thicknesses, are made of various types of plastics, such as polyamide, ABS or acrylic, and these features determine their stiffness. Thanks to their design, they are resistant to water, moisture and sweat. Products equipped with them can be washed without having to remove them from the orthosis. Our plastic stays work only in one direction, perfectly stabilize the laterally protected part of the body, adjusting to it at the same time and have a memory function, thanks to which they always return to their original shape. This function causes the stays in the orthosis to stabilize the swollen limb immediately after the injury and after the swelling has come off. The plastic stays cannot bend and that is why, they cannot correct the body posture or the secured joint.

PADDINGS
3D supports
3D relief supports are independent technical solutions to relieve the rigid elements of a given orthosis. These elements are made of supporting foams or EVA foam. These foams are connected with various types of skin-friendly materials and materials with an adhesive function. These pads have the appropriate shape and color adapted to the type of orthosis. They relieve both metal elements of orthoses, such as splints, stays, underwires and orthopedic drop locks, as well as other elements that should not come into direct contact with the patient's skin. These pads have an anatomical shape and are made of comfortable foam with proper hardness and elasticity, guaranteeing the proper therapeutic effect.
Setting up
Downloads
Accessories
ACCESSORIES / PRODUCTS TO BE USED WITH
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.